Principal investigators
The Lipoprotein(a) Studies Collaboration is jointly led by Peter Willeit (Medical University of Innsbruck) and Sotirios Tsimikas (University of Califonia-San Diego).
Key publication
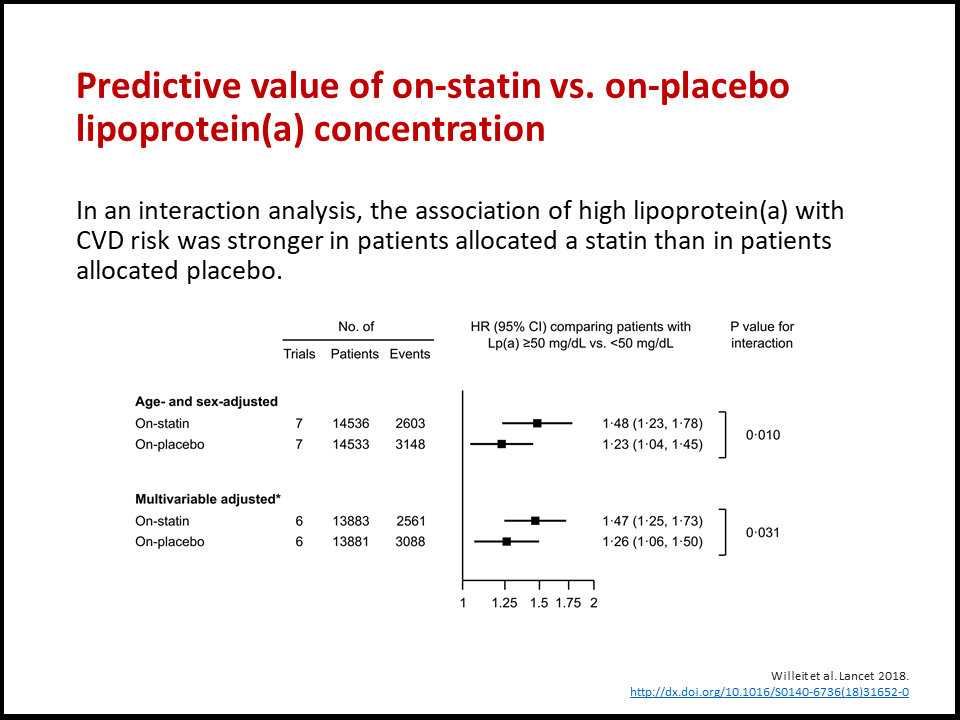
The key publication from this collaboration has been published in the Lancet on 4 October 2018. Please click on the subsequent links to obtain full-text and a slide deck.

Rationale
- Lipoprotein(a) [Lp(a)] is a lipoprotein composed of apolipoprotein(a) covalently bound to apolipoprotein B of a low-density lipoprotein-like particle.
- Lp(a) is an established risk factor for cardiovascular disease. Lp(a) has pro-inflammatory and pro-thrombotic properties and may thereby promote the developement of atherosclerosis.
- In contrast to other major lipoproteins, there is currently no approved specific therapy available to lower circulating plasma levels of Lp(a).
Overarching aim
Lipoprotein(a) Studies Collaboration (LPASC) has harmonised Lp(a) data from prospective cohort studies and clinical trials. The overarching aim of the consortium is to:
- better understand the role of Lp(a) as a independent risk marker in the primary vs. secondary prevention setting,
- characterise the effects of different medications on Lp(a) concentration, and
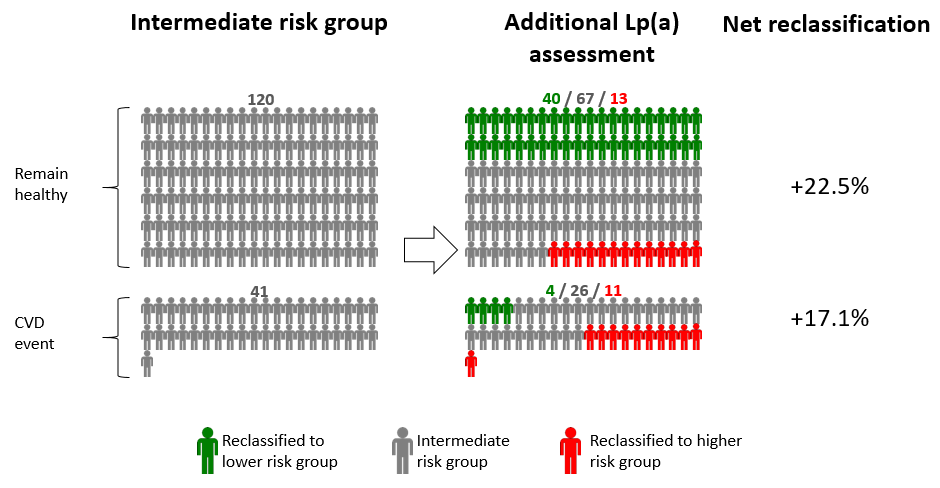
- evaluate the usefulness of Lp(a) measurements in risk prediction models over and beyond information on conventional risk factors.
Collaborating studies
| Cohort | Study design | Years of baseline | Population type | Country |
|---|---|---|---|---|
| 4D | Clinical trial (Atorvastatin) | 1998-2002 | Type 2 diabetes + hemodialysis | Germany |
| 4S | Clinical trial (Simvastatin) | 1989-1990 | Prior myocardial infarction or angina | Scandinavian countries |
| AFCAPS | Clinical trial (Lovastatin) | 1990-1993 | General population | USA |
| ASTRONOMER | Clinical trial (Rosuvastatin) | 2002-2005 | Asymptomatic mild to moderate aortic stenosis | Canada |
| CARDS | Clinical trial (Atorvastatin) | 1997-2001 | Type 2 diabetes | UK & Ireland |
| FHCHILDREN | Clinical trial (Pravastatin) | 1997-1999 | Homozygous familial hypercholesterolemia | The Netherlands |
| JUPITER | Clinical trial (Rosuvastatin) | 2003-2006 | General population with C-reactive protein >2mg/dL | Multinational |
| LIPID | Clinical trial (Pravastatin) | 1990-1992 | Prior myocardial infarction or unstable angina | Australia & NZ |
| MIRACL | Clinical trial (Atorvastatin) | 1997-1999 | Acute coronary syndrome | Multinational |
| PRINCE | Clinical trial (Pravastatin) | 2000 | Primary and secondary prevention | USA |
| PROVE-IT | Clinical trial (Pravastatin & Atorvastatin) | 2000-2001 | Acute myocardial infarction or high-risk unstable angina | Multinational |
| PROXI | Clinical trial (Pravastatin & Atorvastatin) | 2001-2002 | General population | USA |
| REVERSAL | Clinical trial (Pravastatin & Atorvastatin) | 1999-2001 | Coronary stenosis | USA |
| TNT | Clinical trial (Atorvastatin) | 1989-1990 | Coronary heart disease | Multinational |
| VISION | Clinical trial (Pitavastatin & Atorvastatin) | 2006 | Hypercholesterolemia | Japan |
Additional publications